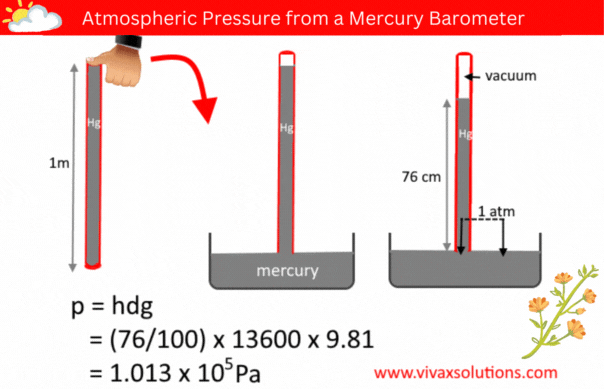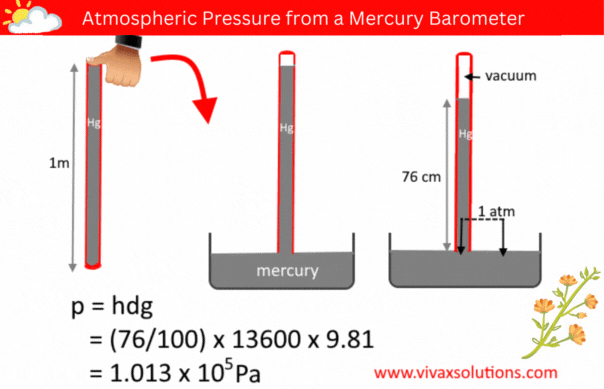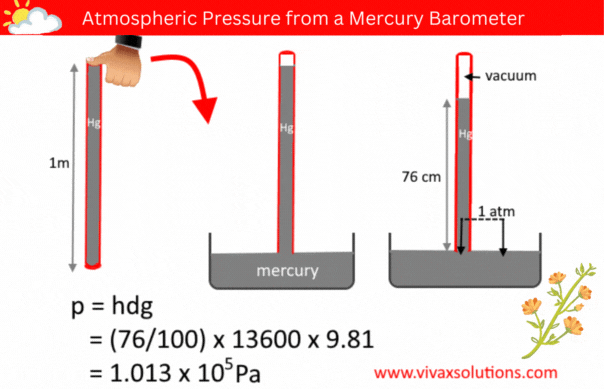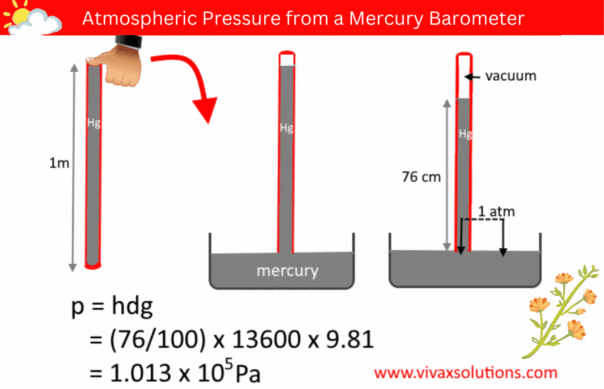
The pressure of a liquid column is given by the formula, p = hdg, where h, d and g are the depth/height, density and the acceleration due to gravity respectively.
A few decades ago, in order to measure the atmospheric pressure, a simple barometer was used. It consisted of a thin, long, glass tube - sealed at one end - a laboratory stand with a clamp to hold it and a container full of mercury.
The glass tube was usually a meter in length. In a school lab, a technician normally used to fill it with mercury to the brim, place his thumb at the open end, before inverting it and placed in the mercury container, while holding the tube vertically with the aid of the stand and clamp.
When left for a while for mercury to settle down, the technician measure the height of the mercury column. If the location in question is at a level closer to the sea level, the height of the mercury column is usually 76 cm.
Since the pressure of a liquid at the same level is equal, the above diagram shows that the pressure of the mercury column is the atmospheric pressure, indeed.
p = hdg
p = (76/100) x 136000 x 9.81
= 1.013 x 105 Pa
Food for thought:
What would happen, if the liquid was water - cheap, less heavy and above all, not toxic at all?
The following, simple calculation shows why water is not suitable:
p = hdg
1.013 x 105 = h x 1000 x 9.81
h = (1.013 x 105) /(1000 x 9.81)
h = 10.3 m
You need a glass tube that is longer than 10 meters - height of more than that of two double decker buses!





0 comments:
Post a Comment